Two years back, Ekta found herself at her dining table in Gurugram, engaged in a call with a representative from the Kiran Patient Access Program at 6:45 PM. She recounted spending over an hour on the phone, hesitant to disconnect, as she awaited a one-time password essential for her mother-in-law to receive her subsequent Keytruda dosage, a prominent immunotherapy drug for cancer, at AIIMS in Delhi the following morning.
For Ajit, a 63-year-old diagnosed with kidney cancer in 2017, the journey to obtaining Keytruda was somewhat smoother, primarily due to his cancer insurance policy. He recalled a challenging moment when a hernia developed at a surgical site following his kidney removal. With his daughter’s wedding approaching, doctors urged him to move the ceremony up, casting doubt on his survival. He has now been receiving Keytruda treatment for over a year.
The experiences of Ekta and Ajit highlight the significant obstacles many face in accessing Keytruda. An investigation by The Indian Express, in collaboration with the International Consortium of Investigative Journalists (ICIJ), has revealed that obtaining Keytruda in India, even through the manufacturer’s patient assistance program, is fraught with financial, logistical, and regulatory hurdles, making the drug largely unavailable to the majority of patients who require it.
On Monday, The Indian Express reported that the restricted access has fueled a growing black market for Keytruda across multiple cities.
Keytruda, known as Pembrolizumab, is produced by the pharmaceutical company Merck & Co (MSD) and is a type of immunotherapy that has proven effective for various forms of cancer. While it is globally approved for numerous cancers, the patient access program in India supports only ten types, including specific lung cancers, head and neck cancer, cervical cancer, renal cancer, and melanoma.
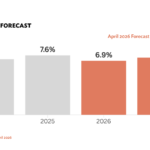
Due to its efficacy, Keytruda has become the best-selling drug worldwide, with sales projected to reach USD 29.5 billion in 2024, representing nearly half of Merck’s total revenue. However, in India, most patients remain unable to access it.
The initial barrier is the access process itself. Without the Kiran Patient Access Program, the market price for the drug exceeds Rs 3 lakh for the recommended 200 mg per infusion. This program offers one complimentary 100 mg vial with each purchased vial for the first five infusions, followed by an additional 25 free vials. However, to qualify, patients must first buy a minimum of five 100 mg vials, costing just under Rs 10 lakh upfront. If they require more, they must re-enroll and pay approximately Rs 10 lakh again.
For Ekta’s family, gathering that amount required a combination of corporate insurance from her sister-in-law’s employer and contributions from her husband and brother-in-law. Conversely, Ajit was able to rely on his specialized insurance plan, which covered the initial Rs 10 lakh for his first doses before he could benefit from the free doses offered through the program.
However, once enrolled in the program, the process remains complicated. Each dose requires the family to submit an infusion form along with a signed and stamped outpatient department (OPD) card that includes the doctor’s prescription. “We often received responses stating that signatures or stamps were missing, necessitating extra trips to AIIMS,” Ekta explained.
A delivery agent would then verify all documents against what was submitted via email. “If we had to take her to the hospital for any complications or side effects between infusion visits and the doctor noted anything on the card, the agent would say it didn’t match the emailed version,” she added.
The program mandates that one registered caregiver must be present at each infusion to collect the medication, which often required Ekta’s husband to take time off work for each treatment cycle.
Her mother-in-law needed a dose every three weeks within a three-day timeframe, as stipulated by the drug’s requirements. Failing to attend an appointment could mean losing the scheduled AIIMS slot, and securing another within the same three days was, according to Ekta, nearly impossible. “The process was burdensome, and if there were any alternative means to obtain the drug, most individuals would likely avoid this route. Yet, it remains challenging for many since it necessitates one person to be completely dedicated to it,” she stated.
Beyond procedural hurdles, The Indian Express investigation identified four structural barriers that hinder access to Keytruda in India.
Firstly, information about the program is scarce. Patients primarily rely on their doctors for details. A Delhi-based oncologist highlighted that this gap can lead to exploitation: “Some individuals offer to provide the drug at significantly reduced prices to desperate patients. However, these drugs are often counterfeit… there is no legitimate way to offer the drug for less than what is available through the patient access program.”
Secondly, the financial threshold to enter the program is prohibitively high for many. Even with the program, the Rs 10 lakh needed for initial doses is too steep. Furthermore, the program excludes patients who earn over Rs 25 lakh annually or have insurance coverage exceeding Rs 25 lakh. “Patients earning more than Rs 25 lakh a year or holding a sum-insured above Rs 25 lakh cannot qualify for the program. Nowadays, there are health insurance plans covering crores of rupees, particularly for cancer treatment. However, high sum coverage can be detrimental for patients,” the oncologist explained.
In numerous instances, doctors noted that registrations are made under the names of lower-earning family members, or alternative coverage with lower sum insured is sought to circumvent the eligibility restrictions. “Even if an individual earns Rs 25 lakh a year, how can they afford a drug costing nearly Rs 4 lakh per infusion every three weeks?” questioned the oncologist.