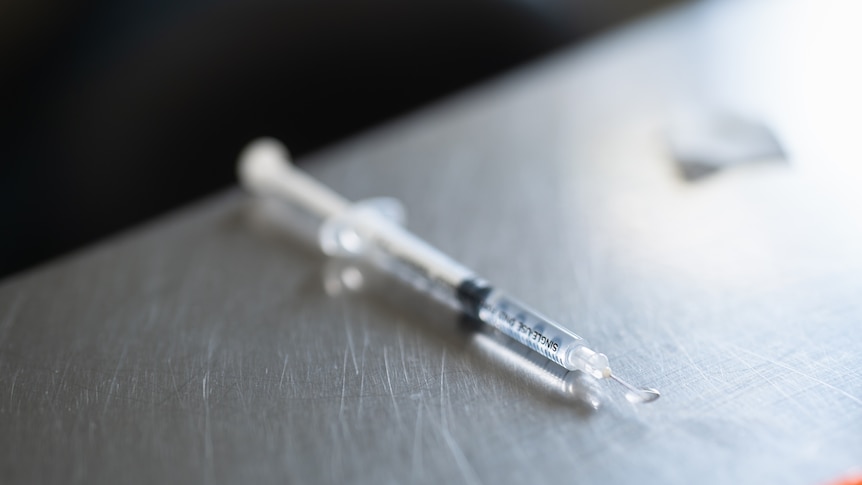
The Therapeutic Goods Administration (TGA) has raised concerns about the dangers associated with unregulated peptides, responding to a notable rise in their use as injectables. The agency has reported instances of “severe allergic reactions” linked to these unauthorized products, prompting calls for stricter regulation.
The TGA has issued a safety warning regarding unapproved peptides, asserting that this emerging wellness trend presents a “substantial” risk to public health. The agency expressed its worries over the increasing import and distribution of these non-approved peptides, which have not undergone rigorous evaluation for safety, quality, or effectiveness.
Unregulated injectable peptides have gained significant traction in Australia, particularly through social media platforms where users share their purported advantages and purchasing tips. The TGA has documented severe allergic reactions resulting in hospitalization, along with symptoms such as intense itching, severe inflammation, heart palpitations, pain, insomnia, blurred vision, and musculoskeletal issues.
In its statement, the TGA emphasized its ongoing efforts to monitor the situation from both safety and enforcement angles. This includes scrutinizing the manufacturing processes, sterility, biological effects, potential side effects, and the actual contents of these products.
Examples of unapproved peptide products include BPC157, GHKCu, TB500, retatrutide, and CJC1295, which are commonly available in injectable formats. The TGA noted that it is illegal to knowingly import, manufacture, or distribute therapeutic goods that lack regulatory approval, including these peptides.
Peptides are short amino acid chains that, when combined, form proteins essential to the body. Many social media users are drawn to these substances due to promises of youthful skin, weight loss, and increased muscle mass, despite their unregulated status and lack of approval for human use.
While several peptide-based medications are authorized by the TGA, they require a doctor’s prescription and supervision. Notable examples of approved peptides include GLP-1 medications like Ozempic and insulin.
Michael Bonning, chair of the Australian Medical Association’s public health committee, stated that the TGA’s advisory serves as a crucial notice for both healthcare professionals and the public. He commended the agency for being more transparent about the risks associated with these products, which have not been assessed for safety or effectiveness.
Dr. Bonning advocated for enhanced regulatory powers for the TGA to proactively address these risks, suggesting the need for more vigorous measures to keep pace with emerging trends.
A TGA representative confirmed that the agency is actively engaged in “compliance and enforcement action” against illegal products, including efforts to eliminate unlawful online advertisements. The representative noted that penalties for serious violations could reach $16.5 million in fines or seven years of imprisonment; however, reports of adverse events associated with unapproved products are not publicly disclosed.
Timothy Piatkowski, a senior research fellow at the University of Queensland studying the risks of certain peptides, welcomed the TGA’s safety alert as a positive development. Nevertheless, he remarked that more action is necessary to catch up with the growing use of these substances.
Dr. Piatkowski called for a comprehensive approach to address the situation, emphasizing the importance of health education, safety information dissemination, and guidance on interpreting online content to help individuals make informed, evidence-based decisions.